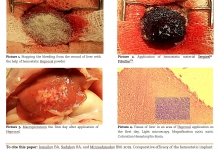
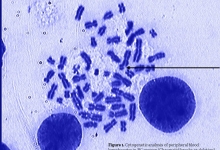
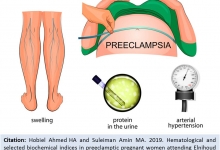
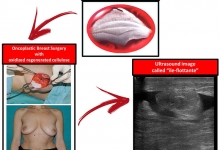
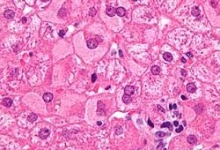
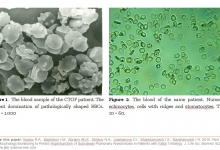
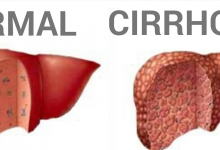
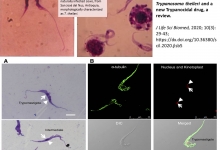
Vancouver Style: The University of Queensland have a modified Vancouver Style on their Library website
More than 6000 styles are available for download from the EndNote Website.
How to install additional styles
1. Download the style (for JLSB reference style click here)
2. Double-click the style file. It should open in EndNote
3. On the open style, go to “File Menu” and choose “Save As”. Replace the word “copy” with your style’s name and click “Save”.
4. Click on “File Menu” and choose “Close Style”
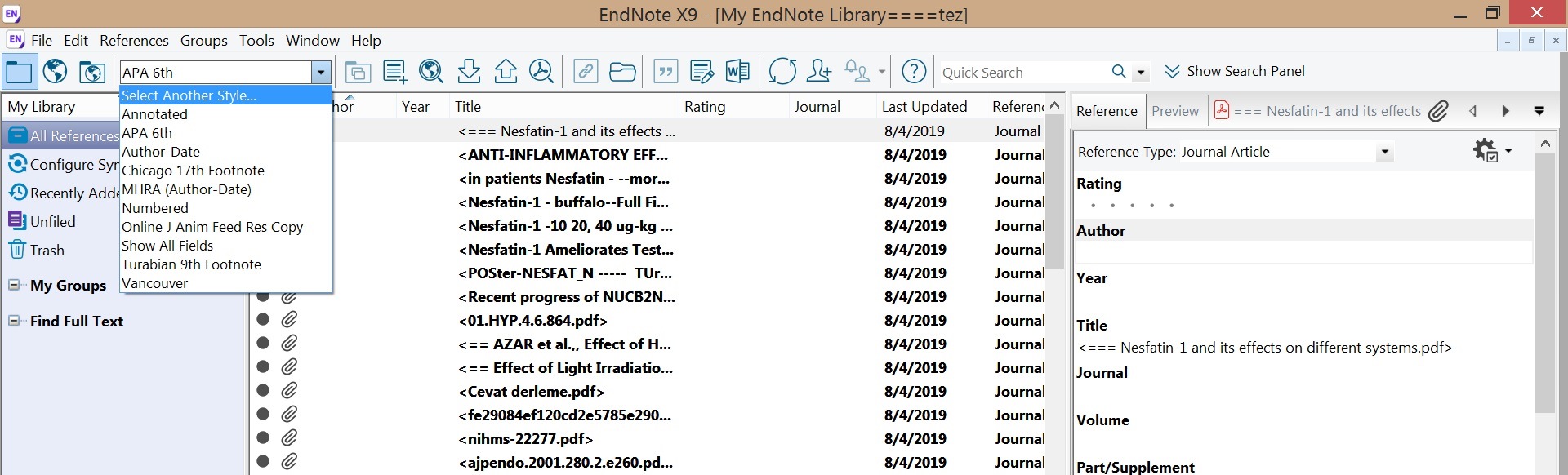
5. Then Go back to your EndNote Library and go to "select another style". Choose it from the list
Where to save your styles
The 400 styles which come with EndNote are usually located in this location:
These can stay here
C:\Program Files\EndNote\Styles
Any extra styles will need to be saved outside of your Program files, into your personal styles folder. EndNote will then merge these folders into one list when you go to choose a style.
To find the correct folder location for your personal styles folder, first open EndNote and look in Edit>Preferences>Folder locations. This will tell you the default file path. The first time you should create the Styles folder following this pattern.
It will look similar to this:
C:\Users\UserName\My Documents\EndNote\Styles
Save any modified styles you download into your personal styles folder
Change Styles
In EndNote, reference or bibliographic style templates are called 'Output Styles'. EndNote supports output styles for in-text citations, numbered in-text citations, and footnotes.
Approx 500 common styles come pre-loaded with EndNote, listed under "Select Another Style".
Choose a Style
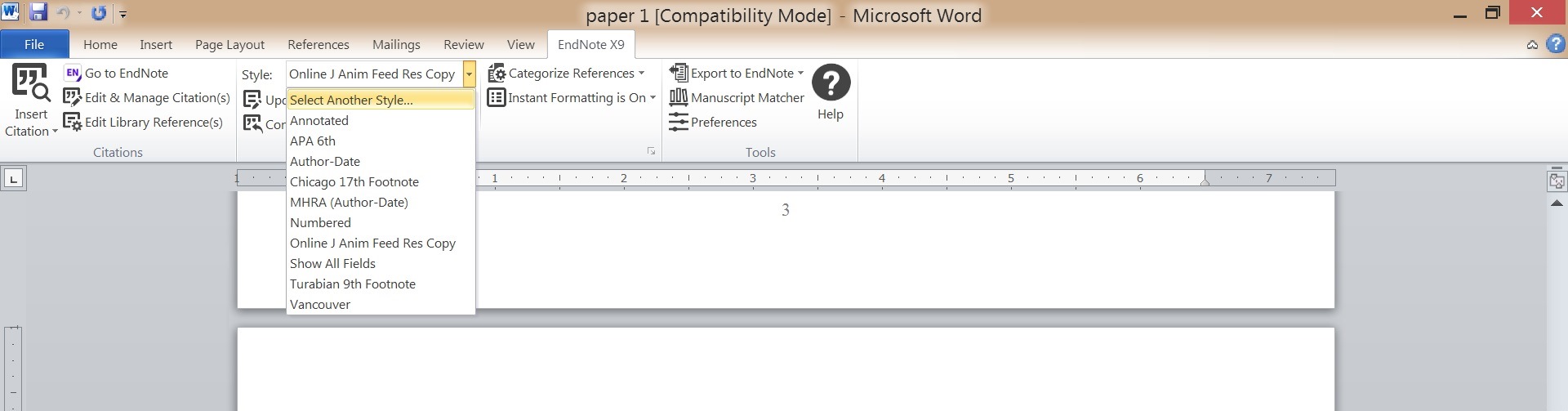
Change Styles in Word
Go to the EndNote Toolbar in Word to change styles and update citations/bibliography in your Word document
Can't find your style in the list?
If the style you just installed does not appear in the drop-down styles menu in EndNote or Word, you need to activate the style by:
- Clicking on Edit in the top toolbar
- Select Styles > Open style manager
- Find and tick the box for the styles you need
- Close the Style manager
- Close and re-open EndNote
- This style will now be available for you to choose
Graphical Abstract
A Graphical Abstract is a single, concise, pictorial and visual summary of the main findings of the article. This could either be the concluding figure from the article or a figure that is specially designed for the purpose, which captures the content of the article for readers at a single glance. Please see the attached examples.
The Graphical Abstract will be displayed in online search result lists, the online contents list and the online article, but will not (yet) appear in the article PDF file or print.
Author instructions
A Graphical Abstract should allow readers to quickly gain an understanding of the main take-home message of the paper and is intended to encourage browsing, promote interdisciplinary scholarship, and help readers identify more quickly which papers are most relevant to their research interests. Authors must provide an image that clearly represents the work described in the paper. A key figure from the original paper, summarising the content can also be submitted as a graphical abstract.
Specifications
A Graphical Abstract should be a one-image file and should visualize one process or make one point clear. For ease of browsing, the Graphical Abstract should have a clear start and end, preferably "reading" from top to bottom or left to right. Try to reduce distracting and cluttering elements as much as possible.
- Image size: Please provide an image with a minimum of 500 x 1300 pixels (hxw) using a minimum resolution of 300 dpi. If you are submitting a larger image then please use the same ratio (200 high x 500 wide).
- Font: Please use Times, Arial, Courier or Symbol font with a large enough font size as the image will be reduced in size for the Table of Contents to fit a window of 200 pixels high.
- File type: preferred file types are TIFF, EPS, PDF or MS Office files.
- No additional text, outline or synopsis should be included. Any text or label must be part of the image file. Please do not use unnecessary white space or a heading “Graphical Abstract” within the image file.
See GA examples
Publisher Item Identifier
A Publisher Item Identifier (PII) is a standard identifier number system which identifies a published document. It was created in 1996, agreed and developed by many of international societies like by the American Chemical Society, the American Institute of Physics, the American Physical Society, Elsevier Science, and the IEEE. Although, PII has currently been included in Digital Object Identifier (DOI)® system (as a syntaxe in the DOI suffix) for Crossref publishers using De facto identifier method, but many of publishers like AMS, still rely on original, facil and free of charge method of creating the Article Unique Identifiers like PII.
Digital Object Identifier (DOI): A name (not a location) for an entity on digital networks. DOI names are used to provide current information, including where they (or information about them) can be found on the Internet.
Publisher Item Identifier (PII): Alphanumeric string of characters that uniquely identifies the article and can be used for future cataloging, searching, and electronic retrieval (Free of Charge). An article explaining the PII numbering system can be found on: http://en.wikipedia.org/wiki/Publisher_Item_Identifier.
-
- 2019 (Volume 9)
- Article 01: S225199391900001-9
- Article 02: S225199391900002-9
- Article 03: S225199391900003-9
- Article 04: S225199391900004-9
- Article 05: S225199391900005-9
- Article 06: S225199391900006-9
- Article 07: S225199391900007-9
- Article 08: S225199391900008-9
- Article 09: S225199391900009-9
- Article 10: S225199391900010-9
- Article 11: S225199391900011-9
- Article 12: S225199391900012-9
- Article 13: S225199391900013-9
- Article 14: S225199391900014-9
- Article 15: S225199391900015-9
- Article 16: S225199391900016-9
- Article 17: S225199391900017-9
- Article 18: S225199391900018-9
- Article 19: S225199391900019-9
- Article 20: S225199391900020-9
- Article 21: S225199391900021-9
- Article 22: S225199391900022-9
- Article 23: S225199391900023-9
- Article 24: S225199391900024-9
- Article 25: S225199391900025-9
- Article 26: S225199391900026-9
- Article 27: S225199391900027-9
- 2020 (Volume 10)
- Article 01: S225199392000001-10
- Article 02: S225199392000002-10
- Article 03: S225199392000003-10
- Article 04: S225199392000004-10
- Article 05: S225199392000005-10
- Article 06: S225199392000006-10
- Article 07: S225199392000007-10
- Article 08: S225199392000008-10
- Article 09: S225199392000009-10
- Article 10: S225199392000010-10
- Article 11: S225199391600031-6
- Article 12: S225199391600032-6
- Article 33: S225199391600033-6
- Article 34: S225199391600034-6
- Article 35: S225199391600035-6
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
- Article 39: S225199391600039-6
- Article 31: S225199391600031-6
- Article 32: S225199391600032-6
- Article 33: S225199391600033-6
- Article 34: S225199391600034-6
- Article 35: S225199391600035-6
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
- 2021 (Volume 11)
- Article 01: S225199392100001-11
- Article 02: S225199392100002-11
- Article 03: S225199392100003-11
- Article 04: S225199392100004-11
- Article 05: S225199392100005-11
- Article 06: S225199392100006-11
- Article 07: S225199392100007-11
- Article 08: S225199392100008-11
- Article 09: S225199392100009-11
- Article 10: S225199392100010-11
- Article 11: S225199392100011-11
- Article 12: S225199392100012-11
- Article 13: S225199392100013-11
- Article 14: S225199392100014-11
- Article 35: S225199391600035-6
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
- Article 39: S225199391600039-6
- Article 31: S225199391600031-6
- Article 32: S225199391600032-6
- Article 33: S225199391600033-6
- Article 34: S225199391600034-6
- Article 35: S225199391600035-6
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
-
- 2016 (Volume 6)
- Article 01: S225199391600001-6
- Article 02: S225199391600002-6
- Article 03: S225199391600003-6
- Article 04: S225199391600004-6
- Article 05: S225199391600005-6
- Article 06: S225199391600006-6
- Article 07: S225199391600007-6
- Article 08: S225199391600008-6
- Article 09: S225199391600009-6
- Article 10: S225199391600010-6
- Article 11: S225199391600011-6
- Article 12: S225199391600012-6
- Article 13: S225199391600013-6
- Article 14: S225199391600014-6
- Article 15: S225199391600015-6
- Article 16: S225199391600016-6
- Article 17: S225199391600017-6
- Article 18: S225199391600018-6
- Article 19: S225199391600019-6
- Article 20: S225199391600020-6
- Article 21: S225199391600021-6
- 2017 (Volume 7)
- Article 01: S225199391700001-7
- Article 02: S225199391700002-7
- Article 03: S225199391700003-7
- Article 04: S225199391700004-7
- Article 05: S225199391700005-7
- Article 06: S225199391700006-7
- Article 07: S225199391700007-7
- Article 08: S225199391700008-7
- Article 09: S225199391700009-7
- Article 10: S225199391700010-7
- Article 11: S225199391700011-7
- Article 12: S225199391700012-7
- Article 13: S225199391700013-7
- Article 14: S225199391700014-7
- Article 15: S225199391700015-7
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
- Article 39: S225199391600039-6
- Article 36: S225199391600036-6
- Article 40: S225199391600040-6
- 2018 (Volume 8)
- Article 01: S225199391800001-8
- Article 02: S225199391800002-8
- Article 03: S225199391800003-8
- Article 04: S225199391800004-8
- Article 05: S225199391800005-8
- Article 06: S225199391800006-8
- Article 07: S225199391800007-8
- Article 08: S225199391800008-8
- Article 09: S225199391800009-8
- Article 10: S225199391800010-8
- Article 11: S225199391800011-8
- Article 12: S225199391800012-8
- Article 13: S225199391800013-8
- Article 14: S225199391800014-8
- Article 15: S225199391800015-8
- Article 36: S225199391600036-6
- Article 37: S225199391600037-6
- Article 38: S225199391600038-6
- Article 39: S225199391600039-6
- Article 36: S225199391600036-6
- Article 40: S225199391600040-6
2015 (Volume 5)
-
- Article 01: S225199391500001-5
- Article 02: S225199391500002-5
- Article 03: S225199391500003-5
- Article 04: S225199391500004-5
- Article 05: S225199391500005-5
- Article 06: S225199391500006-5
- Article 07: S225199391500007-5
- Article 08: S225199391500008-5
- Article 09: S225199391500009-5
- Article 10: S225199391500010-5
- Article 11: S225199391500011-5
- Article 12: S225199391500012-5
- Article 13: S225199391500013-5
- Article 14: S225199391500014-5
- Article 15: S225199391500015-5
- Article 16: S225199391500016-5
- Article 17: S225199391500017-5
- Article 18: S225199391500018-5
- Article 19: S225199391500019-5
- Article 20: S225199391500020-5
- Article 21: S225199391500021-5
- Article 22: S225199391500022-5
- Article 23: S225199391500023-5
- Article 24: S225199391500024-5
- Article 25: S225199391500025-5
- Article 26: S225199391500026-5
- Article 27: S225199391500027-5
- Article 28: S225199391500028-5
- Article 29: S225199391500029-5
- Article 30: S225199391500030-5
- Article 31: S225199391500031-5
- Article 32: S225199391500032-5
- Article 33: S225199391500033-5
- Article 34: S225199391500034-5
- Article 35: S225199391500035-5
- Article 36: S225199391200036-5
- Article 37: S225199391200037-5
- Article 38: S225199391200038-5
- Article 39: S225199391200039-2
- Article 40: S225199391200040-2
.
2014 (Volume 4)
|
|
|
|
|
|
|
|
2013 (Volume 3)
|
|
|
|
2012 (Volume 2)
|
|
|
2011 (Volume 1)
|
![]()
Open Access is a kind of free and unrestricted online availability of knowledge that provides immediate, worldwide, barrier-free access to the full text of research articles without requiring a subscription to the journal in which these articles are published. Based on the original BOAI statement (Budapest Open Access Initiative) we signed (http://www.budapestopenaccessinitiative.org/list_signatures), readers are allowed to copy, use, distribute, transmit and display the work publicly and to make and distribute derivative works, in any digital medium for any responsible purpose, subject to proper attribution of authorship.
Since, an article processing (tracking, editing, formatting, page design, online publication, hosting, etc.) has considerable cost for each journal's team that do not charge for access, they rely on other means of funding publication. In this publishing model, the costs are usually covered by the author’s institution or research funds. These Open Access charges replace subscription charges and allow the publishers to make the published material freely available to all interested online readers and audiences. At the same time, authors who publish in Open Access journals retain the copyright of their article.
Other than their cost-recovery model, Open Access journals are no different from traditional subscription-based journals; they undergo the same peer-review and quality control as any other scholarly journal. Moreover, Open Access allows for maximum visibility, uptake and use of the published material.
![]() Scienceline journals apply the Creative Commons Attribution NonCommercial 4.0 License (CC BY-NC 4.0) to all manuscripts to be published.
Scienceline journals apply the Creative Commons Attribution NonCommercial 4.0 License (CC BY-NC 4.0) to all manuscripts to be published.
An Open Access Publication is one that meets the following two conditions:
1. The author(s) and copyright holder(s) grant(s) to all users a free, irrevocable, worldwide, perpetual right of access to, and a license to copy, use, distribute, transmit and display the work publicly and to make and distribute derivative works, in any digital medium for any responsible purpose, subject to proper attribution of authorship, as well as the right to make small numbers of printed copies for their personal use.
2. A complete version of the work and all supplemental materials, including a copy of the permission as stated above, in a suitable standard electronic format is deposited immediately upon initial publication in at least one online repository that is supported by an academic institution, scholarly society, government agency, or other well-established organization that seeks to enable open access, unrestricted distribution, interoperability, and long-term archiving.
Authors who publish with this journal agree to the following terms: 1. Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal. 2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal. 3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
| JLSB EndNote Style |
| Manuscript Template (.doc) |
| Sample Articles |
| Declaration form |
| Policies and Publication Ethics |
Table of Contents:
ARTICLE TYPES
PREPARATION OF MANUSCRIPT
Article sections format
Declarations
REFERENCES
Review/Decisions/Processing
The Journal of Life Science and Biomedicine publishes papers of clinical interest in life science and biomedicine. Manuscripts may be submitted in the form of original articles, review articles, case reports, letters to the Editor and guidelines... view full aims and scope. The journal aims to provide its readers with papers of the highest quality and impact through a process of peer review and editorial work. Duties and responsibilities of all the subjects involved in the editorial process are summarized at Publication ethics. Manuscripts are expected to comply with the instructions to authors which conform to the Uniform Requirements for Manuscripts Submitted to Biomedical Editors by the International Committee of Medical Journal Editors (ICMJE).
Submission of manuscripts
Papers should be submitted directly to the online Editorial Office via one of the following SCIENCELINE Online Submission System or JLSB Online Submission Form. We do consider only online submissions, but not multiple submissions, so please send only one manuscript at a time, either by JLSB or Scienceline Submission Forms. Do not send a second submission until you have heard about the first. Simultaneous submissions to other journals are also not allowed.
Please embed all figures and tables in the manuscript to become one single file for submission. Once submission is complete, the system will generate a manuscript ID and will send an email regarding your submission. The authors can track submissions via This email address is being protected from spambots. You need JavaScript enabled to view it.; This email address is being protected from spambots. You need JavaScript enabled to view it.. All manuscripts must be checked by English native speaker and submitted in English for evaluation (in totally confidential and impartial way).
The journal does not apply any charges for online submission. All authors are responsible for their research. Authors are requested to choose a corresponding author who is responsible for the following requirements: managing all communications between the journal and all co-authors during the manuscript submission, peer review, publication process and after publication; ensuring that the names of authors, their arrangement and affiliations are correct; ensuring that all listed authors have approved the manuscript before submission; making sure all permissions to reproduce previously published material have been obtained from the copyright owner; making sure disclosures, declarations, statements from all authors are included in the manuscript as appropriate.
Authors are welcome to suggest 2 unbiased expert reviewers when they submit their manuscript by providing their names, institutions and e-mail addresses. Authors should make sure that the suggested reviewers have a high degree of expertise and independence in the field of the study presented. Please note that suggestions are welcome and may help facilitate the peer-review process but the journal will not guarantee to use them.
Supplementary information
The JLSB online submission form allows supplementary information to be submitted together with the main manuscript file and covering letter. If you have more than one supplementary files, you can also submit the extra ones by email after the initial submission. Author guidelines are specific for each journal. Our MS Word template can assist you by modifying your page layout, text formatting, headings, title page, image placement, and citations/references such that they agree with the guidelines of journal. If you believe your article is fully edited per journal style, please use our MS Word template before submission. Supplementary materials may include figures, tables, methods, videos, and other materials. They will available online in JLSB website linked to the original published article. Supplementary tables and figures should be labeled with a "S", e.g. "Table S1" and "Figure S1". The maximum file size for supplementary materials is 10MB each. Please keep the files as small possible to avoid the frustrations experienced by readers with downloading large files.
ETHICAL RESPONSIBILITY OF AUTHORS
Submission of the manuscript means that it is original and has not been published or under evaluation elsewhere, is not plagiarized and does not infringe any copyright or right of privacy. If accepted, the manuscript will not be published elsewhere either wholly or in part in any form or language except in case of specific agreements. The manuscript must be approved by all co-authors, if any via signing the declaration form (.docx). Specific discipline rules should be followed by authors for acquiring, selecting and processing data. Results should be presented clearly, honestly and without fabrication or inappropriate data manipulation.
Statement of human rights
All articles reporting studies that involving human subjects must include a statement at the methods section of the manuscript, clearly stating that the study has been approved by the institutional research ethics committee before experiment was started (including the name of the board/committee or its chairperson, the protocol number and the date of approval that was attributed by the ethics committee) and that has been conducted in accordance with the principles of Helsinki Declaration.
Patient consent
Authors should include at the beginning of the methods section of their manuscript a statement clearly indicating that patients have given their informed consent for participation in the research study and enough precaution has been taken to protect the privacy of patients. Authors should obtain permission from the patients for the publication of photographs or other material that might identify them, otherwise, the face of all the patients should be covered or the face part should be cut in the images. If necessary, a copy of such permission may be requested.
Statement on welfare of animals
Authors of experiments on animals should include a statement at the methods section of the manuscript indicating that the study was approved by the institutional research ethics committee and specifying the guidelines for care of animals (Guidelines for the Treatment of Animals in Research and Teaching) that have been followed.
Graphical abstract
 Authors should provide a graphical abstract (a beautifully designed feature figure) to represent the paper aiming to catch the attention and interest of readers. Graphical abstract will be published online in the table of content. The graphical abstract should be colored, and kept within an area of 12 cm (width) x 6 cm (height) or with similar format. Image should have a minimum resolution of 300 dpi and line art 1200dpi.
Authors should provide a graphical abstract (a beautifully designed feature figure) to represent the paper aiming to catch the attention and interest of readers. Graphical abstract will be published online in the table of content. The graphical abstract should be colored, and kept within an area of 12 cm (width) x 6 cm (height) or with similar format. Image should have a minimum resolution of 300 dpi and line art 1200dpi.
Note: Height of the image is generally no more than the width. Authors should avoid putting too much information into the graphical abstract as it occupies only a small space. The graphical abstract can be presented in the format of PowerPoint, Word, PDF, JPG, PNG or TIFF, after a manuscript is accepted for publication. For preparing a Professional Graphical Abstract, please click here.
ARTICLE TYPES
Instructions for the most frequent types of articles submitted to the journal.
Original articles that should be original contributions to the subject. The text can be 3000-5500 words not including references, tables, figures. No more than 45 references will be accepted, except for the critical articles like COVID-19. The article must be subdivided into the following sections: introduction, materials and methods, results, discussion, conclusions. A TIDIeR checklist (http://www.equator-network.org/reporting-guidelines/tidier) should be included in the covering letter where an intervention is described. The Journal supports the efforts to increase quality of writing in scientific papers. In this case, we suggest authors to conform the structure of their paper to the checklist requirements of one of the following guidelines and add in the covering letter the relevant checklist:
- randomized controlled trials (CONSORT - http://www.consort-statement.org);
- non-randomized controlled trials (TREND - http://www.cdc.gov/trendstatementa);
- observational studies (STROBE - http://www.strobe-statement.org);
- quality improvement in health care (SQUIRE - http://www.squire-statement.org).
Reviews and meta-analyses. The text should be 6000-12000 words not including references, tables, figures. No more than 100 references will be accepted, except for the critical articles. The article must be subdivided into the following sections: introduction, results, discussion, conclusions. In this case, we suggest authors to conform the structure of their paper to the checklist requirements of one of the following guidelines and add in the covering letter the relevant checklist:
- systematic reviews (PRISMA - http://www.prisma-statement.org);
- meta-analyses of observational studies (MOOSE - http://www.equatornetwork.org/index.aspx?0=1052).
Case reports that give a description of particularly interesting cases. The text should be 1500-2000 words not including references, tables, figures. No more than 10 references will be accepted, except for the critical articles. The article must be subdivided into the following sections: introduction, case report or clinical series, discussion, conclusions. In this case, we suggest authors to conform the structure of their paper to the checklist requirements of the following guidelines and add in the covering letter the relevant checklist:
- CARE for case reports (http://www.care-statement.org).
Letters to the Editor may refer to articles already published in the journal or to particularly interesting observations or scientific data that the authors wish to present to readers in a concise form (for example please see the Letter of Prof. Dr. Franceschini et al., 2019). The text is acceptable in both simple or subdivided forms and should be 500-1000 words not including references, tables, figures. No more than 7 references will be accepted, except for the critical articles.
Main format
First page of the manuscripts must be properly identified by the title and the name(s) of the author(s). It should be typed in Times New Roman or Zilla Slab (font sizes: 17pt for the title, 10pt for the section headings in the body of the text and the main text, double spaced, in A4 format with 2cm margins (both doc./docx formats). All pages and lines of the main text should be numbered consecutively throughout the manuscript. Abbreviations in the article title are not allowed. Manuscripts should be arranged in the following order:
1. TITLE (brief, attractive and targeted);
2. Name(s) and Affiliation(s) of author(s) (including post code and corresponding Email);
3. ABSTRACT;
4. Key words (select from MeSH terms, separate by comma,);
5. Abbreviations (those used throughout the manuscript, if required);
6. INTRODUCTION (clear statement of the problem, the relevant literature on the subject, and the proposed approach or solution);
7. MATERIAL AND METHOD (should be complete enough to allow experiments to be reproduced);
8. RESULTS;
9. DISCUSSION; Results and Discussion can be presented jointly.
10. CONCLUSION; Discussion and Conclusion can be presented jointly.
11. DECLARATIONS (Acknowledgements, Consent to publish, Competing interests, Authors' contributions, and Availability of data etc.)
12. REFERENCES;
13. Tables;
14. Figures;
15. Graphs
Title should be a brief phrase describing the contents of the paper. The first letter of each word in title should use upper case. The Title Page should include the author(s)'s full names and affiliations, the name of the corresponding author along with phone and e-mail information. Present address (es) of author(s) should appear as a footnote.
Abstract should be informative and completely self-explanatory, briefly present the topic, state the scope of the experiments, indicate significant data, and point out major findings and conclusions. The best length for the abstract is 250 words; However for long article or with high volume data it should be no longer than 300 words in length. Complete sentences, active verbs, and the third person should be used, and the abstract should be written in the past tense. Standard nomenclature should be used and abbreviations should be avoided. No literature should be cited.
Following the abstract, about 3 to 8 key words that will provide indexing references should be listed. In order to assist indexers in cross-indexing articles please use keywords from the Medical Subject Headings (MeSH) list of Index Medicus (http://www.nlm.nih.gov/mesh/MBrowser.html).
Introduction should provide a clear statement of the problem, the relevant literature on the subject, and the proposed approach or solution. It should be understandable to colleagues from a broad range of scientific disciplines.
Material and Method should be complete enough to allow experiments to be reproduced. However, only truly new procedures should be described in detail; previously published procedures should be cited, and important modifications of published procedures should be mentioned briefly. Capitalize trade names and include the manufacturer's name and address. Subheadings should be used. Methods in general use need not be described in detail. The ethical approval for using human and animals in the researches should be indicated in this section with a separated title.
Results should be presented with clarity and precision. The results should be written in the past tense when describing findings in the author(s)'s experiments. Previously published findings should be written in the past tense. Results should be explained, but largely without referring to the literature. In case of the effectiveness of a particular drug or other substances as inhibitor in biological or biochemical processes, the results should be provided as IC50 (half maximal inhibitory concentration) or similar appropriate manner.
Discussion should interpret the findings in view of the results obtained in this and in past studies on this topic. State the conclusions in a few sentences at the end of the paper. The Results and Discussion sections can include subheadings, and when appropriate, both sections can be combined.
Conclusion should be brief and tight about the importance of the work or suggest the potential applications and extensions. This section should not be similar to the Abstract content.
Declarations including Acknowledgements, Author contribution, Competing interests, Consent to publish, and Availability of data etc.
Tables should be kept to a minimum and be designed to be as simple as possible. Tables are to be typed double-spaced throughout, including headings and footnotes. Each table should be on a separate page, numbered consecutively in Arabic numerals and supplied with a heading and a legend. Tables should be self-explanatory without reference to the text. The details of the methods used in the experiments should preferably be described in the legend instead of in the text. The same data should not be presented in both table and graph forms or repeated in the text.
Figure legends should be typed in numerical order on a separate sheet. Graphics should be prepared using applications capable of generating high resolution GIF, TIFF, JPEG or PowerPoint before pasting in the Microsoft Word manuscript file. Use Arabic numerals to designate figures and upper case letters for their parts (Figure 1). Begin each legend with a title and include sufficient description so that the figure is understandable without reading the text of the manuscript. Information given in legends should not be repeated in the text.
Declarations
Please ensure that the sections: Ethics (and consent to participate, if any), Acknowledgements, Author contribution, Competing interests, Consent to publish, Availability of data and materials are included at the end of your manuscript in a Declarations section.
Acknowledgements
We encourage authors to include an Acknowledgements section. Please acknowledge anyone who contributed towards the study by making substantial contributions to conception, design, acquisition of data, or analysis and interpretation of data, or who was involved in drafting the manuscript or revising it critically for important intellectual content, but who does not meet the criteria for authorship. Please also include their source(s) of funding. Please also acknowledge anyone who contributed materials essential for the study. Authors should obtain permission to acknowledge from all those mentioned in the Acknowledgements. Please list the source(s) of funding for the study, for each author, and for the manuscript preparation in the acknowledgements section. Authors must describe the role of the funding body, if any, in study design; in the collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
Author contribution
For manuscripts with more than one author, JLSB require an Author Contributions section to be placed after the Acknowledgements section. An 'author' is generally considered to be someone who has made substantive intellectual contributions to a published study. To qualify as an author one should 1) have made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; 2) have been involved in drafting the manuscript or revising it critically for important intellectual content; and 3) have given final approval of the version to be published. Each author should have participated sufficiently in the work to take public responsibility for appropriate portions of the content. Acquisition of funding, collection of data, or general supervision of the research group, alone, does not justify authorship.
We suggest the following format/example (please use initials to refer to each author's contribution): AB carried out the molecular genetic studies, participated in the sequence alignment and drafted the manuscript. JY carried out the immunoassays. MT participated in the sequence alignment. ES participated in the design of the study and performed the statistical analysis. FG conceived of the study, and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
For authors that equally participated in a study please write 'All/Both authors contributed equally to this work.' Contributors who do not meet the criteria for authorship should be listed in an acknowledgements section.
Competing interests
Competing interests that might interfere with the objective presentation of the research findings contained in the manuscript should be declared in a paragraph heading "Competing interests" (after Acknowledgment or Author Contribution sections). Examples of competing interests are ownership of stock in a company, commercial grants, board membership, etc. If there is no competing interest, please use the statement "The authors declare that they have no competing interests.".
Journal of Life Science and Biomedicine adheres to the definition of authorship set up by The International Committee of Medical Journal Editors (ICMJE). According to the ICMJE authorship criteria should be based on 1) substantial contributions to conception and design of, or acquisition of data or analysis and interpretation of data, 2) drafting the article or revising it critically for important intellectual content and 3) final approval of the version to be published. Authors should meet conditions 1, 2 and 3. It is a requirement that all authors have been accredited as appropriate upon submission of the manuscript. Contributors who do not qualify as authors should be mentioned under Acknowledgements.
Consent to publish
Please include a ‘Consent for publication section in your manuscript. If your manuscript contains any individual person’s data in any form (including individual details, images or videos), consent to publish must be obtained from that person, or in the case of children, their parent or legal guardian. All presentations of case reports must have consent to publish. You can use your institutional consent form or our consent form if you prefer. You should not send the form to us on submission, but we may request to see a copy at any stage (including after publication). If your manuscript does not contain any individual persons data, please state “Not applicable” in this section.
Change in authorship
We do not allow any change in authorship after provisional acceptance. We cannot allow any addition, deletion or change in sequence of author name. We have this policy to prevent the fraud.
Data deposition
Nucleic acid sequences, protein sequences, and atomic coordinates should be deposited in an appropriate database in time for the accession number to be included in the published article. In computational studies where the sequence information is unacceptable for inclusion in databases because of lack of experimental validation, the sequences must be published as an additional file with the article.
JLSB accept the manuscripts in PDF, Word or TeX/LaTeX formats; Word files are preferred especially those are prepared using EndNote®. However, our team will reformat the articles of non-EndNote users via EndNote in the alley proof stage, if accepted.
.
A JLSB reference style for EndNote may be found here.
How to install additional styles? Please click here.
How to turn on "Jumping" from a citation to the bibliography? Please click here.
.
We prefer Vancouver referencing style that is often used in medicine and the natural sciences. Uniform requirements for manuscripts submitted to Biomedical Journals, published by International Committee of Medical Journal Editors, includes a list with examples of references https://www.nlm.nih.gov/bsd/uniform_requirements.html in the Vancouver style. It is desirable to add a DOI (digital object identifier) number for each reference. If a DOI is lacking, please add a link to any online source of an article (Google Scholar is preferred).
- References should be numbered consecutively and cited in the text by number in square brackets like [1, 2] instead of parentheses (and not by author and date).
- References should not be formatted as footnotes. Avoid putting personal communications and unpublished observations as references, as much as possible. Papers that have been accepted for publication, but not yet specified for an issue should be cited as “to be published”. Papers that have been submitted for publication should be cited as “submitted for publication”.
- All references to publications made in the text should be presented in a list with their full bibliographical description. DOI number or the link of article should be added to the end of the each reference. All the cited papers in the text must be listed in References and all the papers in References must be cited in the text; To this end, we suggest you use our EndNote style.
- Names of authors and title of journals, published in non-latin alphabets should be transliterated in English, as much as possible. Please give the English citation first, followed by the original foreign-language citation.
- Capitalize only the first word in a paper title, except for proper nouns and element symbols.
- A sample of standard reference is "1th Author surname A, 2th Author surname B and 3th Author surname C. Article title should be regular and 9 or 10 pt. Journal of Life Science and Biomedicine or J. Life Sci. Biomed. 2020; Volume(Issue): 00-00." "DOI: https://dx.doi.org/10.00000/xxxx"; PMID: 000000; PMCID: PMC000000; "Google Scholar (https://scholar.google.com)"
- To find DOI of each reference easily, click here (https://apps.crossref.org/SimpleTextQuery); To find the reference with the acceptable format in PubMed click here (https://pubmed.ncbi.nlm.nih.gov/).
- From 2020, the journal titles should be full in references. The titles should not be italic.
- References with more than 6 authors should be listed, the first 6 authors followed by 'et al.'
- Use a space after authors’ initials.
- The color of references in the text of article is blue. Example: [1, 2].
- At least 35% of the references of any submitted manuscript (for all types of article) should include scientific results published in the last five years.
-Examples (at the text- blue highlighted)
Smit [1] stated that ...; Smit and Janak [2] believe that …; Our results are in agreement with previous studies [3, 4] or study of Nurai et al. [5].
Acceptable Examples (at references section):
For Journals:
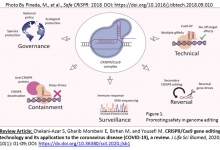
1. Chekani-Azar S, Gharib Mombeni E, Birhan M, and Yousefi M. CRISPR/Cas9 gene editing technology and its application to the coronavirus disease (COVID-19), a review. Journal of Life Science and Biomedicine. 2020; 10(1): 01-09; DOI: https://dx.doi.org/10.29252/scil.2020.jlsb1
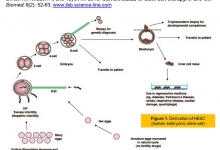
2. Nazirov FG, Djuraeva NM, Vakhidova NT, Omonov OA and Salimov UR. Diagnostic informativity of the volume MDCT-angiography and MR-cholangiography in the pre- and intraoperative periods for the examination of donors of a liver fragment. J. Life Sci. Biomed., 2019; 9(6): 151-156. DOI: https://dx.doi.org/10.36380/scil.2019.jlsb24
3. Doll MA, Salazar-González RA, Bodduluri S and Hein DW. Arylamine N-acetyltransferase 2 genotype-dependent N-acetylation of isoniazid in cryopreserved human hepatocytes. Acta Pharm Sin B. 2017; 7(4):517-522. DOI: https://dx.doi.org/10.00000/xxxx; Link
4. Titomanlio L, Kavelaars A, Dalous J, Mani S, El Ghouzzi V, et al. Stem cell therapy for neonatal brain injury: Perspectives and challenges. Annals of neurology. 2011; 70 (5): 698-712. DOI: https://dx.doi.org/10.00000/xxxx; Link
5. Karen KS and Otto CM. Pregnancy in women with valvular heart disease. Heart. 2007 May; 93(5): 552–558. DOI: https://dx.doi.org/10.00000/xxxx; Link
For In press manuscripts (maximum 2)
Hasan V, Sri Widodo M and Semedi B. Oocyte Diamater Distribution and Fecundity of Javaen Barb (Systomus Orphoides) at the Start of Rainy Season in Lenteng River, East Java, Indonesia insurance. In press. 2015.
For symposia reports and abstracts
Cruz EM, Almatar S, Aludul EK and Al-Yaqout A. Preliminary Studies on the Performance and Feeding Behaviour of Silver Pomfret (Pampus argentens euphrasen) Fingerlings fed with Commercial Feed and Reared in Fibreglass Tanks. Asian Fisheries Society Manila, Philippine. 2000; 13: 191-199.
For Conference
Skinner J, Fleener B and Rinchiuso M. Examining the Relationship between Supervisors and Subordinate Feeling of Empowerment with LMX as A Possible Moderator. 24th Annual Conference for Industrial Organizational Behavior. 2003.
For Book
Russell, Findlay E. Snake Venom Poisoning, 163, Great Neck, NY: Scholium International. ISBN 0-87936-015-1. 1983.
For Web Site
Bhatti SA and Firkins JT. 2008. http://www.ohioline.osu.edu/sc1156_27.hmtl.
Nomenclature and Abbreviations
Nomenclature should follow that given in NCBI web page and Chemical Abstracts. Standard abbreviations are preferable. If a new abbreviation is used, it should be defined at its first usage. Abbreviations should be presented in one paragraph, in the format: "term: definition". Please separate the items by ";".
E.g. ANN: artificial neural network; CFS: closed form solution; ...
Abbreviations of units should conform with those shown below:
|
Decilitre
|
dl
|
Kilogram
|
kg
|
|
Milligram
|
mg
|
hours
|
h
|
|
Micrometer
|
mm
|
Minutes
|
min
|
|
Molar
|
mol/L
|
Mililitre
|
ml
|
|
Percent
|
%
|
.
|
Other abbreviations and symbols should follow the recommendations on units, symbols and abbreviations: in “A guide for Biological and Medical Editors and Authors (the Royal Society of Medicine London 1977). Papers that have not been published should be cited as “unpublished”. Papers that have been accepted for publication, but not yet specified for an issue should be cited as “to be published”. Papers that have been submitted for publication should be cited as “submitted for publication".
Formulae, numbers and symbols
- Typewritten formulae are preferred. Subscripts and superscripts are important. Check disparities between zero (0) and the letter 0, and between one (1) and the letter I.
- Describe all symbols immediately after the equation in which they are first used.
- For simple fractions, use the solidus (/), e.g. 10 /38.
- Equations should be presented into parentheses on the right-hand side, in tandem.
- Levels of statistical significance which can be used without further explanations are *P < 0.05, **P < 0.01, and ***P < 0.001.
- In the English articles, a decimal point should be used instead of a decimal comma.
- Use Symbol fonts for "±"; "≤" and "≥" (avoid underline).
- In chemical formulae, valence of ions should be given, e.g. Ca2+ and CO32-, not as Ca++ or CO3.
- Numbers up to 10 should be written in the text by words. Numbers above 1000 are recommended to be given as 10 powered x.
- Greek letters should be explained in the margins with their names as follows: Αα - alpha, Ββ - beta, Γγ - gamma, Δδ - delta, Εε - epsilon, Ζζ - zeta, Ηη - eta, Θθ - theta, Ιι - iota, Κκ - kappa, Λλ - lambda, Μμ - mu, Νν - nu, Ξξ - xi, Οο - omicron, Ππ - pi, Ρρ - rho, Σσ - sigma, Ττ - tau, Υυ - ipsilon, Φφ - phi, Χχ - chi, Ψψ - psi, Ωω - omega.Please avoid using math equations in Word whenever possible, as they have to be replaced by images in xml full text.
.
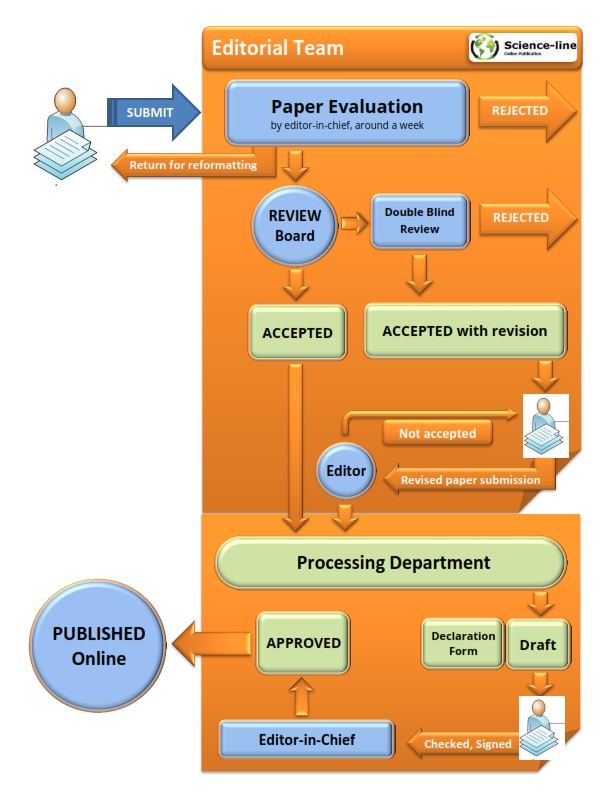
Firstly, all manuscripts will be checked by Docol©c and or PlagScan, plagiarism finding tools. The received papers with plagiarism rate of more than 15% for approved research papers (main text not including references, tables, figures) and 25% for approved review papers will be rejected. Manuscripts that are judged to be of insufficient quality or unlikely to be competitive enough for publication will be returned to the authors at the initial stage. The remaining manuscripts go through a double blind peer-review process by 2 reviewers (from both suggested or internal reviewers) selected by section or deputy editors of JLSB, who are research workers specializing in the relevant field of study. We always try to avoid delays in the reviewing process, but it relies on the time and cooperation of the referees that works without any remuneration, hence, it may take 2 weeks to 2 months. One unfavorable review means that the paper will not be published and possible decisions are: accept as is, minor revision, major revision, or reject. The corresponding authors should submit back their revisions within 14 days in the case of minor revision, or 30 days in the case of major revision.
The authors can resubmit a revision via email (This email address is being protected from spambots. You need JavaScript enabled to view it.) or online submission systems provided that they have checked "Revised" box and mentioned the article code (for example JLSB-1108) in the submission form. Manuscripts with significant results are typically reviewed and published at the highest priority. After review and editing the article, a final formatted proof is sent to the corresponding author once again to apply all suggested corrections during the article process. The editor who received the final revisions from the corresponding authors shall not be hold responsible for any mistakes shown in the final publication.
Plagiarism
There is an instant policy towards plagiarism (including self-plagiarism) in our journal. Manuscripts are screened for plagiarism by Docol©c and PlagScan, before or during publication, and if found they will be rejected at any stage of processing.
Declaration
After manuscript accepted for publication, a declaration form will be sent to the corresponding author who that is responsible to coauthors' agreements to publication of submitted work in JLSB after any amendments arising from the peer review.
Date of issue
All accepted articles are published bi-monthly around 25th of January, March, May, July, September and November, each year in full text on the Internet.
Publication charges
The publication costs are covered through article processing charges (APCs) and No submission fee, or any other processing fees are required for the publication of the accepted article. There is a modest APC of 100 Euro(€) editor fee for the processing of each primary accepted paper (1000-4000 words) to encourage high-quality submissions. APCs are only charged for articles that pass the pre-publication checks and are ready to be published. A surcharge will be placed on any article that is over 4000 words in length to cover the considerable additional processing costs. Payment can be made by credit card, bank transfer, money order or check. Instruction for payment is sent during publication process as soon as manuscript is accepted. Meanwhile, this journal encourages the academic institutions in low-income countries to publish high quality scientific results, free of charges. If the authors have decided to withdraw the accepted manuscript while they have received one of the reviewed or edited versions of manuscript from the journal, they will be banned from the submission for a period of 5 years.
| WORD COUNT | PRICE* |
| 1000-4000 words |
€100 |
| over 4000 words |
€120 |
* The prices are valid until 30th December 2025.
The Waiver policy
The publication fee will be waived for invited authors, authors of hot papers, and corresponding authors who are editorial board members of the Journal of Life Science and Biomedicine. The Journal will consider requests to waive the fee for cases of financial hardship (for high quality manuscripts and upon acceptance for publication). Requests for waiver of the publication fee must be submitted via individual cover letter by the corresponding author and cosigned by an appropriate institutional official to verify that no institutional or grant funds are available for the payment of the fee. Letters including the manuscript title and manuscript ID number should be sent to: This email address is being protected from spambots. You need JavaScript enabled to view it.. It is expected that waiver requests will be processed and authors will be notified within 24 h.
The OA policy
The Journal of Life Science and Biomedicine is an open access journal which means that all content is freely available without charge to the user or his/her institution. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author. This is in accordance with the BOAI definition of Open Access. For additional information please see Policies and Publication Ethics.
.
Paper Submission Flow
 Submission Preparation Checklist
Submission Preparation Checklist
Authors are required to check off their submission's compliance with all of the following items, and submissions may be returned to authors that do not adhere to the following guidelines.
The submission has not been previously published, nor is it before another journal for consideration (or an explanation has been provided in Comments to the Editor).
The submission file is in Microsoft Word, RTF, or PDF document file format.
Where available, URLs for the references have been provided.
The text is single-spaced; uses a 12-point font; and all illustrations, figures, and tables are placed within the text at the appropriate points, rather than at the end.
The text adheres to the stylistic and bibliographic requirements outlined in the Author Guidelines.
(Revised on 25 November 2021)
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.